|
Stellar classification is the categorisation of stars based on their characteristic electromagnetic spectra. In astronomical spectroscopy, the strength, shape, and position of absorption and emission lines, as well as the overall spectral energy distribution of the continuum, reveal many properties of astronomical objects. the frequency of the tuner, it would be the frequency spectrum of the antenna signal. If we made a graph of the strength of each channel vs. The radio then uses a tuned circuit or tuner to select a single channel or frequency band and demodulate or decode the information from that broadcaster. Any particular radio receiver will detect a single function of amplitude (voltage) vs. When many broadcasters are present, the radio spectrum consists of the sum of all the individual channels, each carrying separate information, spread across a wide frequency spectrum. Each broadcast radio and TV station transmits a wave on an assigned frequency range, called a channel. The microwave spectrum corresponds to frequencies between 300 MHz (0.3 GHz) and 300 GHz and wavelengths between one meter and one millimeter. The radio spectrum is the part of the electromagnetic spectrum corresponding to frequencies lower below 300 GHz, which corresponds to wavelengths longer than about 1 mm. In radio and telecommunications, the frequency spectrum can be shared among many different broadcasters. Therefore, flat-line spectra in general are often referred to as white, whether they represent light or another type of wave phenomenon (sound, for example, or vibration in a structure). When all the visible frequencies are present equally, the perceived color of the light is white, and the spectrum is a flat line. A graph of the intensity plotted against the frequency (showing the brightness of each color) is the frequency spectrum of the light. A rainbow, or prism, sends these component colors in different directions, making them individually visible at different angles. Light from many different sources contains various colors, each with its own brightness or intensity. The emission spectrum refers to the spectrum of radiation emitted by the compound due to electron transitions from a higher to a lower energy state. The absorption spectrum of a chemical element or chemical compound is the spectrum of frequencies or wavelengths of incident radiation that are absorbed by the compound due to electron transitions from a lower to a higher energy state. The wavelength of visible light ranges from 390 to 700 nm. The visible spectrum is the part of the electromagnetic spectrum that can be seen by the human eye. Devices used to measure an electromagnetic spectrum are called spectrograph or spectrometer. Spectrum is also used to refer to a graphical representation of the signal as a function of the dependent variable.Įlectromagnetic spectrum refers to the full range of all frequencies of electromagnetic radiation and also to the characteristic distribution of electromagnetic radiation emitted or absorbed by that particular object. The term now applies to any signal that can be measured or decomposed along a continuous variable, such as energy in electron spectroscopy or mass-to-charge ratio in mass spectrometry. It has also been expanded to more abstract " signals", whose power spectrum can be analyzed and processed. 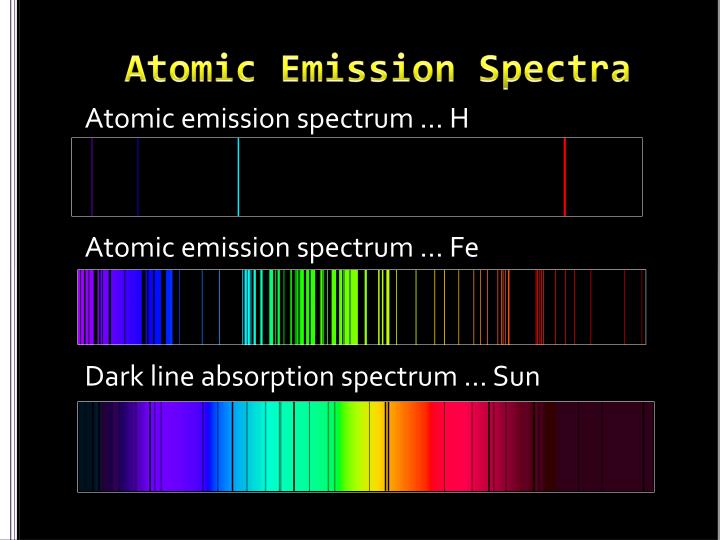
Later it expanded to apply to other waves, such as sound waves and sea waves that could also be measured as a function of frequency (e.g., noise spectrum, sea wave spectrum). Soon the term referred to a plot of light intensity or power as a function of frequency or wavelength, also known as a spectral density plot. 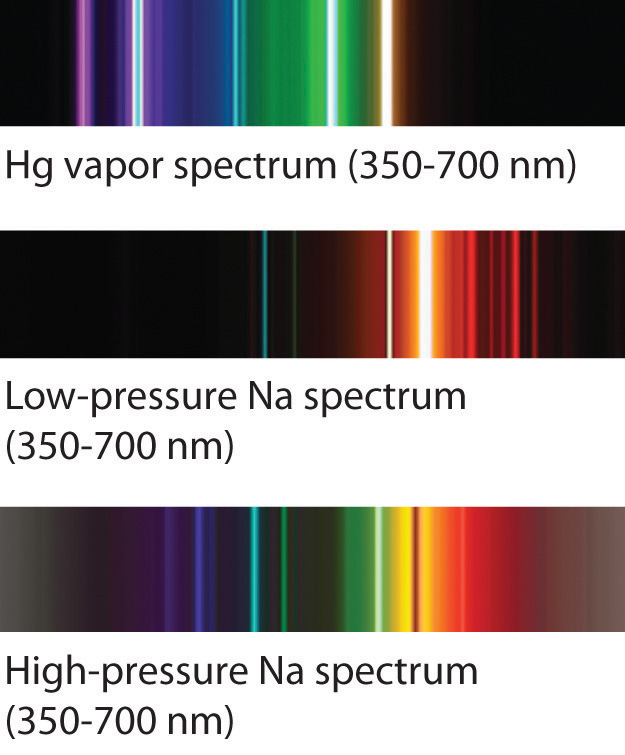
In the physical sciences, the term spectrum was introduced first into optics by Isaac Newton in the 17th century, referring to the range of colors observed when white light was dispersed through a prism. ( August 2022) ( Learn how and when to remove this template message)ĭiagram illustrating the electromagnetic spectrum Please help to improve this article by introducing more precise citations. When exploding fireworks, you will see the color when the metal atoms absorb energy from the detonater.This article includes a list of general references, but it lacks sufficient corresponding inline citations.(CC BY-NC-SA 3.0 Christopher Auyeung via CK-12 Foundation)
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
